this post was submitted on 03 Mar 2026
551 points (100.0% liked)
History Memes
2381 readers
5 users here now
A place to share history memes!
Rules:
-
No sexism, racism, homophobia, transphobia, assorted bigotry, etc.
-
No fascism (including tankies/red fash), atrocity denial or apologia, etc.
-
Tag NSFW pics as NSFW.
-
Follow all Piefed.social rules.
-
History referenced must be 20+ years old.
Banner courtesy of @setsneedtofeed@lemmy.world
OTHER COMMS IN THE HISTORYVERSE:
- !historymusic@quokk.au
- !historygallery@quokk.au
- !historyruins@piefed.social
- !historyart@piefed.social
- !historyartifacts@piefed.social
- !historyphotos@piefed.social
founded 11 months ago
MODERATORS
you are viewing a single comment's thread
view the rest of the comments
view the rest of the comments
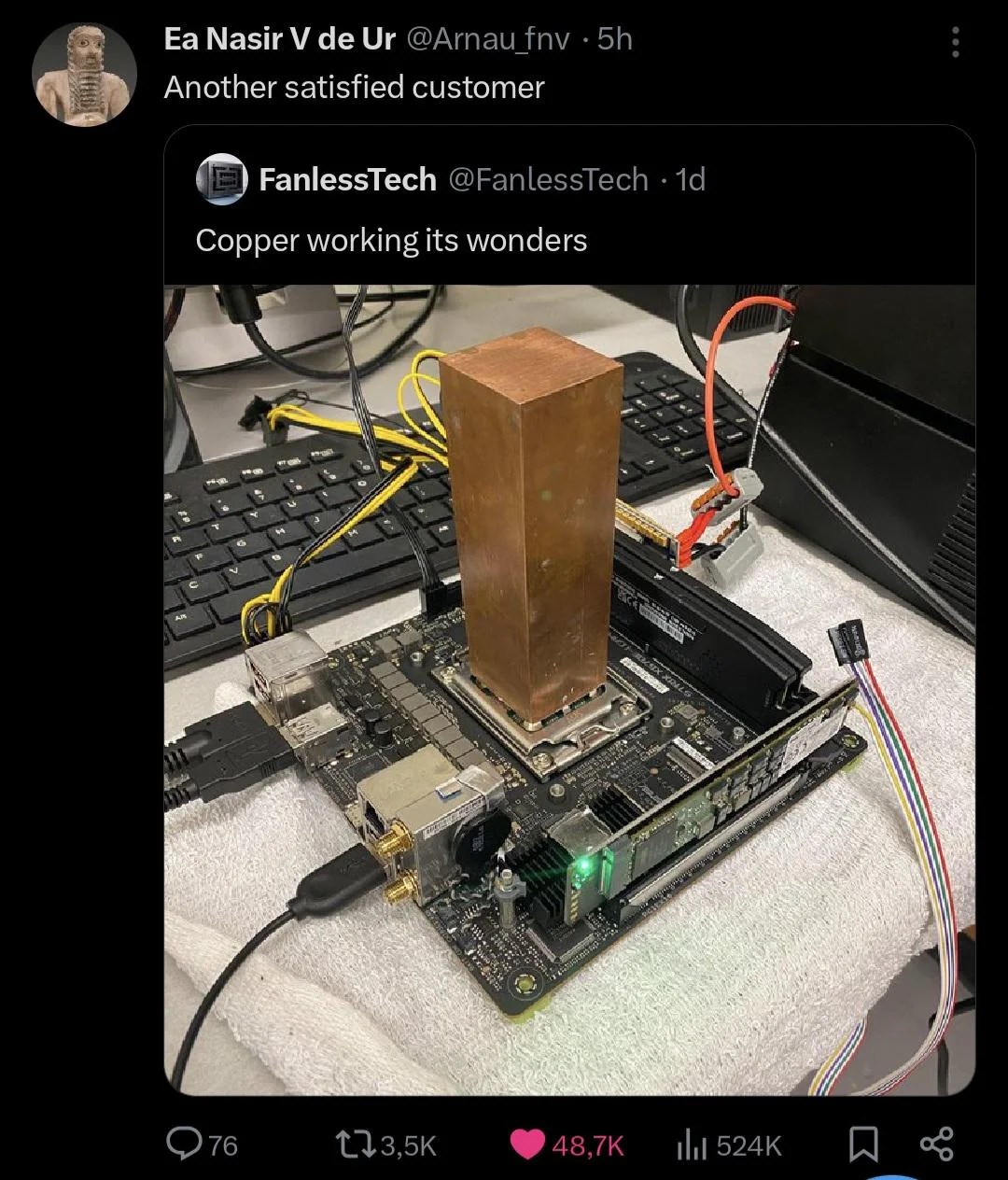
Copper has more mass, heat capacity, and thermal conductivity per litre.
Is aluminium actually more effective as a dissipation surface? I hadn't heard that.
Copper is better conductor but it's worse at dissipation. Do the experience yourself, heat a block of each and then touch them afterwards.
Is that not because the copper holds more heat, so stays hot for longer at the same dissipation?
Maybe you're right, but I remember than in the 2000s I've had identical cpu heatsinks in both copper/aluminum versions, and the aluminum one had better performance. And then they started to make hybrid ones, stating that the copper part was to allow rapid heat transference, and the aluminum part to improve dissipation. But maybe its all marketing.
Aluminium is significantly cheaper, that's why they make coolers with a copper base and alu fins. It's a good compromise.
I just thought of another reason for using Al this way.
Since the heat is being transferred to air, which would be much slower than the 2, using larger fins with lesser thermal conductivity might be more desirable than smaller fins with higher conductivity.
This would also be accompanied with other design changes like greater fin-fin gap, which is better for use with lower pressure fans.
So overall cheaper design and lesser noise.
Aluminium is cheaper and lighter.
This seems to suggest that the metal-air transmission is virtually identical between the two, and cites some sources: https://electronics.stackexchange.com/questions/255731/copper-or-aluminum-heatsink
Dissipation = thermal conductivity. Copper is better in both, it's just heavier and far more expensive. Are you sure you put the same amount of energy into both blocks there? A copper heatsink can generally be much smaller than an aluminium one.
The problems and complications with your method: